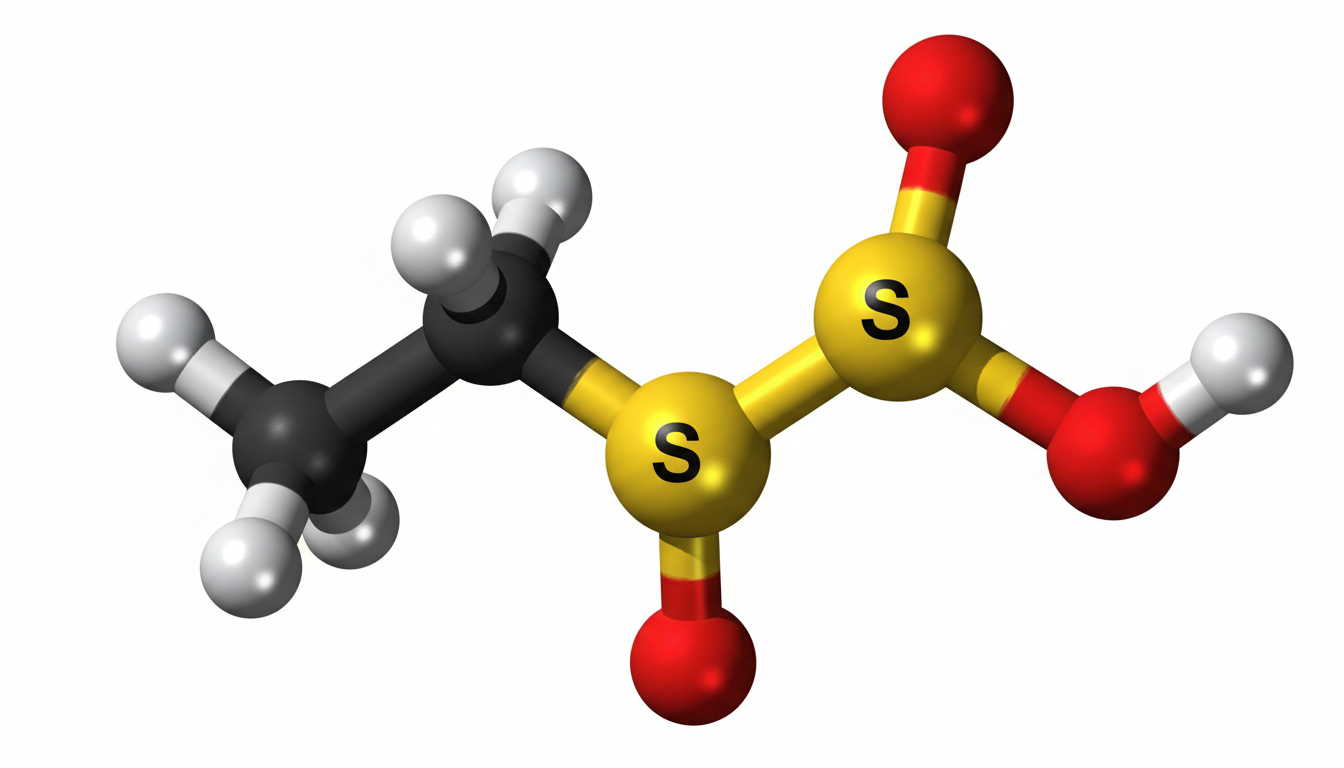
Ethylene sulfate is an important organic compound widely studied in chemistry due to its unique structure and industrial relevance. It belongs to the group of sulfate esters and is primarily known for its reactive properties. Scientists and researchers often explore ethylene sulfate in laboratories for its role in chemical synthesis and reactions. Despite not being a household name, it plays a significant role in specialized industrial and research settings.
Chemical Structure and Composition
Ethylene sulfate has a cyclic structure consisting of an ethylene group bonded to a sulfate group. This cyclic sulfate ester gives it distinct chemical behavior compared to other sulfate compounds. The molecular formula of ethylene sulfate is C2H4O4S, and it features strong polar bonds that contribute to its reactivity. Its structure allows it to participate in various chemical transformations, making it valuable in organic synthesis.
Physical Properties of Ethylene Sulfate
Ethylene sulfate typically appears as a colorless to pale yellow liquid under standard conditions. It has a relatively high boiling point compared to simple organic compounds due to its molecular interactions. The compound is soluble in water and many organic solvents, which enhances its usability in chemical reactions. Its stability can vary depending on environmental conditions such as temperature and exposure to moisture.
Chemical Behavior and Reactivity
One of the most notable characteristics of ethylene sulfate is its high reactivity. It can undergo ring-opening reactions, which are commonly used in organic chemistry to produce other compounds. This reactivity makes it a useful intermediate in chemical processes. However, the same property also requires careful handling, as it can react with various substances under certain conditions.
Industrial Applications of Ethylene Sulfate
Ethylene sulfate is used in several industrial processes, particularly in the production of specialty chemicals. It serves as an intermediate in the synthesis of polymers, pharmaceuticals, and other organic compounds. Industries value it for its ability to facilitate complex chemical reactions efficiently. Although its use is somewhat specialized, it remains an essential component in certain manufacturing sectors.
Role in Organic Synthesis
In organic chemistry, ethylene sulfate is often used as a reagent for introducing sulfate groups into molecules. It is particularly useful in laboratory settings where precise chemical modifications are required. Researchers utilize its reactive nature to study reaction mechanisms and develop new compounds. Its versatility makes it a valuable tool for chemists working in both academic and industrial environments.
Safety and Handling Considerations
Handling ethylene sulfate requires strict safety precautions due to its reactive and potentially hazardous nature. It can cause irritation to the skin, eyes, and respiratory system if not handled properly. Protective equipment such as gloves, goggles, and lab coats is essential when working with this compound. Proper ventilation and adherence to safety guidelines are also crucial to minimize risks.
Environmental Impact of Ethylene Sulfate
The environmental impact of ethylene sulfate depends on how it is used and disposed of. Improper disposal can lead to contamination of water and soil, posing risks to ecosystems. Therefore, industries must follow environmental regulations to ensure safe handling and disposal. Research is ongoing to develop safer alternatives and reduce the ecological footprint of such chemicals.
Storage and Stability Guidelines
Ethylene sulfate should be stored in a cool, dry, and well-ventilated area away from incompatible substances. Proper storage conditions help maintain its stability and reduce the risk of unwanted reactions. Containers should be tightly sealed to prevent exposure to moisture and air. Following recommended storage practices is essential for ensuring safety and preserving the compound’s quality.
Comparison with Other Sulfate Esters
Compared to other sulfate esters, ethylene sulfate stands out due to its cyclic structure. This structural difference gives it unique reactivity and makes it more suitable for certain chemical processes. While other sulfate esters may be more stable, ethylene sulfate offers advantages in terms of versatility and efficiency in reactions. This comparison highlights its importance in specialized applications.
Research and Scientific Importance
Ethylene sulfate continues to be an area of interest in scientific research. Chemists study its properties to better understand reaction mechanisms and develop innovative materials. Its role in advancing chemical knowledge cannot be overlooked. Ongoing research aims to improve its applications while addressing safety and environmental concerns.
Advantages and Limitations
Like any chemical compound, ethylene sulfate has both advantages and limitations. Its high reactivity and versatility make it valuable in various applications. However, its potential hazards and environmental impact require careful management. Balancing these factors is essential for maximizing its benefits while minimizing risks.
Future Prospects of Ethylene Sulfate
The future of ethylene sulfate lies in continued research and innovation. Scientists are exploring new ways to use this compound in sustainable and efficient processes. Advances in green chemistry may lead to safer methods of handling and utilizing ethylene sulfate. As industries evolve, its role may expand into new areas of application.
Conclusion
Ethylene sulfate is a significant chemical compound with diverse applications in research and industry. Its unique structure and reactivity make it an essential tool for chemists. However, proper safety measures and environmental considerations are crucial when working with this compound. Understanding its properties, uses, and risks helps ensure its effective and responsible utilization.